As existing healthcare provider organizations merge and affiliate to create Consolidated Enterprises, image acquisition workflows are often found to be different across the various facilities. Often, the different facilities that comprise the Consolidated Enterprise had different procedures and standard of practice for image acquisition and Quality Control (QC), along with different information and imaging systems.
Standardizing and harmonizing enterprise-wide policies, especially for imaging exam QC, can have significant benefits. A failure to standardize these workflows in a Consolidated Enterprise may result in inconsistent or inaccurate imaging records, which can lead to reading and viewing workflow challenges. These are compounded with a shared imaging system, such as an enterprise PACS or VNA, and can result in delays in care and patient safety risks.
There are generally two areas worth evaluating for optimization:
- Technologist imaging exam acquisition workflow (Tech Workflow)
- Imaging record Quality Control workflow (QC Workflow)
Here, we will explore Tech Workflow. QC Workflow will be covered in a subsequent post.
Throughout this discussion the term Radiology Information System (RIS) is used, which can be a standalone system or a module of an EMR.
Tech Workflow
The use of DICOM Modality Worklist (DMWL) for the management of image acquisition is well-understood and broadly adopted. However, the process of marking an exam as “complete” (or “closed”) following acquisition is less standardized and varies across different vendors and healthcare enterprises. The subsequent QC and diagnostic reading workflows rely on the “completion” of the exam before they can begin. For example, an exam that is never marked as “complete” may not appear on a Radiologist Reading Worklist, and an imaging exam that is marked as “complete” when it isn’t will be available for Radiologists to read with only a partial set of images.
Imaging Technologists typically interact with the following applications on a daily-basis.

- Modality Console – a comprehensive set of tools, attached to the modality, to perform image acquisition activities (such as DMWL queries, exam protocoling, post-processing, etc.).
- Radiology Information System (RIS) – a specific view into the enterprise RIS application, allowing Technologists to look up patient/procedure information, a set of tools to document the acquisition and mark exam as “complete”, etc.
- Image Manager/Archive (IM/A) QC – a comprehensive set of imaging exam Quality Control (QC) tools, provided by the Image Manager/Archive (IM/A), such as PACS or VNA, or a dedicated application, to make any necessary corrections to ensure the quality of acquired imaging exam records.
As stated above, there is significant variability among healthcare providers with respect to instituting Tech Workflow policies and procedures. The following diagram illustrates the steps involved in a common Tech Workflow.
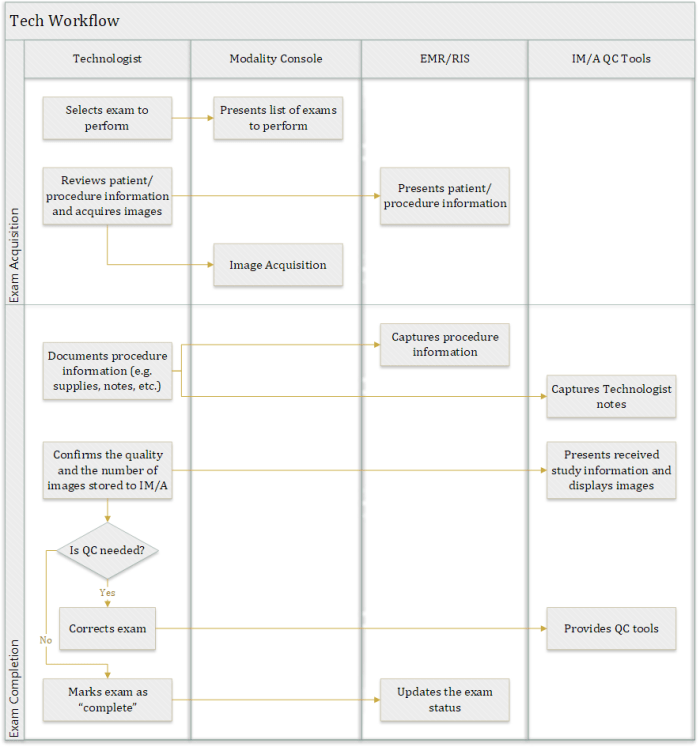
Notes:
- In some cases, Technologists validate the quality of the image and confirm that the number of images in the IM/A is correct for multiple studies at a time instead of each one independently due to the high-volume of exams being acquired.
- An ability to assess the quality of the imaging exam and correct it (if needed) in a quick and user-friendly manner is critical for an efficient exam completion workflow.
PACS-driven Reading Workflow
In this scenario, the PACS Client provides a Reading Worklist and it is typically responsible for launching (in-context, through a desktop integration) the Report Creator application. There are several methods used across provider organizations to communicate study complete status updates to the PACS.
| Method |
Benefit |
Challenge |
| Time out – this is the most typical approach, which considers a study to be complete after a defined period of time has passed (for example, five minutes) since the receipt (by PACS) of the last DICOM object from the modality. |
|
If the time-out is too long, the creation of the corresponding Reading Worklist item will be delayed. Alternatively, a short time-out may result in a Radiologist reporting an incomplete study, which requires follow-up review and potentially an addendum to the report once the missing images are stored to PACS. |
| HL7 ORM – some organizations release HL7 ORM messages to the Report Creator only after the order status is updated (to study complete) in the RIS. |
- Easy to implement
- Prevents reporting of incomplete exams (although relies on Technologists to validate the completeness of the study structure in the PACS)
|
There are scenarios where PACS has received DICOM studies, but their statuses in the RIS application has not yet been updated (for example, as can happen with mobile modalities). The Reading Worklist is unaware of the HL7 message flow between the RIS and the Report Creator and, therefore, allows the Radiologist to start reviewing cases. However, these cases have no corresponding procedure information in the Report Creator. When the Radiologists tries to launch the reporting application in the context of the current study, the Report Creator is unable to comply. |
| DICOM MPPS – Once an exam is complete, a DICOM MPPS N-Set message (issued by the modality) informs the PACS (and/or RIS) about the structure of the study and the fact that it is completed (along with other useful exam information). |
- Prevents reporting of incomplete exams
- Automatic confirmation of the structure of the study
|
- The adoption of DICOM Modality Performed Procedure Step (MPPS) is still limited in most enterprises, even though some modalities, RIS, and PACS support it.
- Somewhat complex to implement (requires integration and testing between each modality and the MPPS server) coupled with a lack of understanding as to the benefits of this approach in many healthcare provider organizations.
- Some modality vendors charge an additional fee for a license to enable MPPS integration.
- Can be disconnected from the “completion” of the exam in RIS (i.e. can ensure the Report Creator’s readiness), provided only the PACS receives and processes the MPPS messages.
|
| DICOM Storage Commitment – Once the exam is complete, a series of DICOM messages (N-Action, N-Event-Report) between modalities and PACS can determine whether a complete study was stored to PACS. |
- Prevents reporting of incomplete exams
- Automatic confirmation of the structure of the study
|
- Although most PACS and many modalities support this DICOM transaction, it is not widely implemented by healthcare providers.
- Somewhat complex to implement (requires integration and testing between each modality and the PACS server) coupled with a lack of understanding as to the benefits of this approach in many healthcare provider organizations.
- Can be disconnected from the “completion” of the exam in RIS (i.e. can ensure the Report Creator’s readiness).
|
RIS-driven Reading Workflow
In this scenario, the RIS provides the Reading Worklist and it is implicitly aware of the status of the exam (assuming the same system is used by Techs and Rads). It creates the worklist item that corresponds to the exam once it reaches the “complete” status. As the Reading Worklist launches both the Report Creator and the Diagnostic Viewer (PACS Client) applications, it does not face the informatics challenges inherent to the PACS-driven Reading Workflow described above.
Enterprise-wide Reading Workflow (Dedicated, Standalone Application)
Some organizations use an enterprise-wide Reading Worklist that is a separate application from the PACS and RIS to orchestrate enterprise-wide diagnostic reading (and other imaging related) tasks across all their Radiologists using fine-grained task-allocation rules. Similar to the RIS-driven Reading Workflow, the worklist launches both the Report Creator and the Diagnostic Viewer applications once a worklist item is selected.
To prevent the complexity of the PACS-driven Reading Workflow described above, some organizations choose to release an HL7 ORM message from the RIS application to the worklist only when the status of the corresponding exam in that system is updated. Alternatively, organizations that choose to send all ORM messages to the worklist application as soon as procedures are scheduled, need to deal with ensuring that the PACS has a complete study prior to allowing it to be reported.
So, what?
It is important for healthcare provider organizations to understand the relationship between the Tech Workflow and the Reading Worklist approach they adopt. If a RIS-driven approach is not chosen, then there should be a clear integration strategy in place to ensure that studies are not reported too soon or missed.





